Current location:Home > Markets > Nutraceuticals


|
Biochem/physiol Actions |
Stigmasterol possesses anti-inflammatory anti-hypercholestrolemic, antitumor and antioxidant functionality. It plays a crucial role in the activation of plasma membrane H+-ATPase and cell proliferation. Variations in stigmasterol and its precursor are noticeable at both the seed and whole plant developmental stages. Stigmasterol may be involved in gravitropism and tolerance to abiotic stress. |
|
Purification Methods |
Stigmasterol is best purified via the tetrabromide-acetate. The impure sterol (3g) is acetylated with Ac2O (60mL) by refluxing for 1.5hour. The mixture is cooled at 20o for 1hour, and the crude acetate is collected. The acetate (3g) in Et2O (30mL) is then treated with Br2/AcOH (38mL, from 5g Br2 in 100mL AcOH), and after cooling at 6o overnight, the tetrabromoacetate is filtered off and washed with Et2O. After six recrystallisations from CHCl3/MeOH the tetrabromoacetate has m 194-196o. This product (1g) in AcOH (12mL) and Zn dust (1g) is refluxed for 1.5hours, filtered hot, diluted with H2O (30mL) and extracted with Et2O. The extract is washed with dilute aqueous sodium sulfite, then H2O, the extract is dried (Na2SO4) and the stigmasterol acetate (~550mg) is recrystallised (4x) from EtOH and twice from MeOH/CHCl3 (2:1) to give the acetate with m 139-148o. This acetate (400mg) is hydrolysed in boiling 10% alcoholic KOH (1mL) for 1hour. Then H2O (30mL) is added and the mixture is extracted with Et2O. The extract is washed with aqueous Na2CO3, then H2O, the solvent is distilled off and the residue is recrystallised (3x) from 95% EtOH to give ~110mg of pure stigmasterol. It is dried in a vacuum over P2O5 for 3hours at 90o. The purity is checked by NMR. The acetate crystallises from MeOH with m 145o, [] D 25 -56o (c 2, CHCl3). [Byerrum & Ball Biochemical Preparations 7 86 1959, Thornton et al. J Am Chem Soc 62 2006 1940, Colin et al. Anal Chem 51 1661 1979, Beilstein 6 IV 4170.] |
|
Definition |
ChEBI: A 3beta-sterol that consists of 3beta-hydroxystigmastane having double bonds at the 5,6- and 22,23-positions. |
|
General Description |
Stigmasterol is a plant sterol that comprises an unsaturated bond between C22 and C23. It is found in foods like margarines and yogurts. Stigmasterol is synthesized from the mevalonate pathway and is present during development stages in plants. |
InChI:InChI=1/C29H48O/c1-7-21(19(2)3)9-8-20(4)25-12-13-26-24-11-10-22-18-23(30)14-16-28(22,5)27(24)15-17-29(25,26)6/h8-9,19-22,24-27H,7,10-18H2,1-6H3/b9-8+/t20-,21-,22-,24+,25-,26+,27+,28+,29-/m1/s1
The present invention discloses novel me...
One compound stigmasteryl-3β-arachidate ...
The methanol soluble part of the concent...
Two new steroidal glycosides (1 and 2) h...
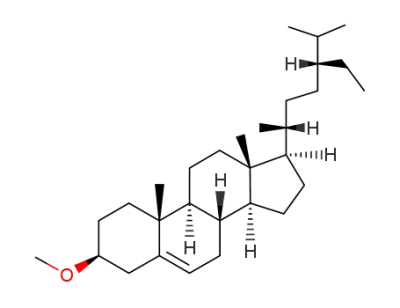
22,23-dihydrostigmasteryl methyl ether

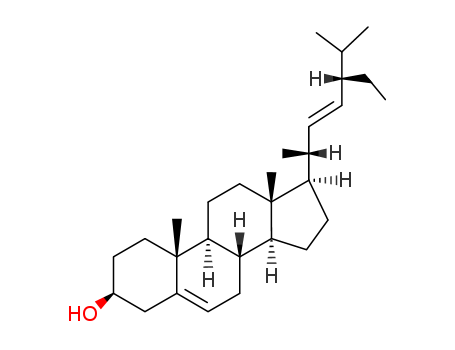
stigmasterol
| Conditions | Yield |
|---|---|
|
With
toluene-4-sulfonic acid;
In
1,4-dioxane; water;
at 100 ℃;
for 2h;
|
90% |
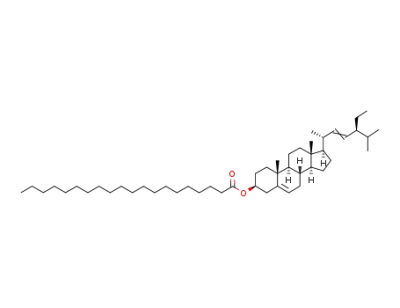
stigmasteryl-3β arachidate

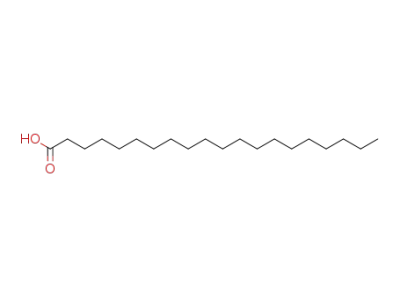
Arachidic acid


stigmasterol
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride; water;
In
1,4-dioxane;
for 4h;
Reflux;
|
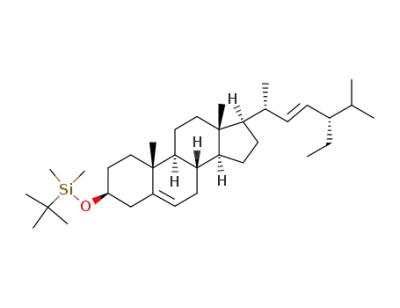
tert-butyl(((3S,8S,9S,10R,13R,14S,17R)-17-((2R,5S,E)-5-ethyl-6-methylhept-3-en-2-yl)-10,13-dimethyl-2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-3-yl)oxy)dimethylsilane
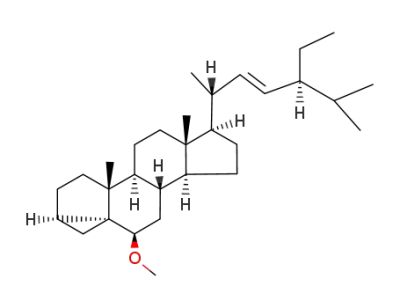
stigmasterol methyl ether
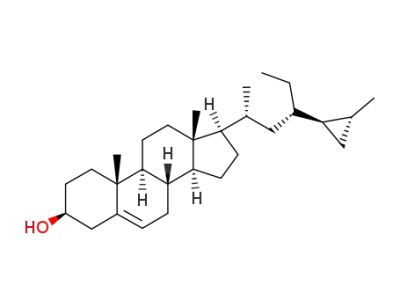
(23S,24R,25R)-23-ethyl-24,26-cyclocholest-5-en-3β-ol
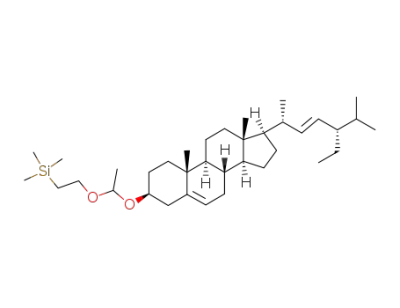
(2-{1-[(3S,8S,9S,10R,13R,14S,17R)-17-((E)-(1R,4S)-4-Ethyl-1,5-dimethyl-hex-2-enyl)-10,13-dimethyl-2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-3-yloxy]-ethoxy}-ethyl)-trimethyl-silane
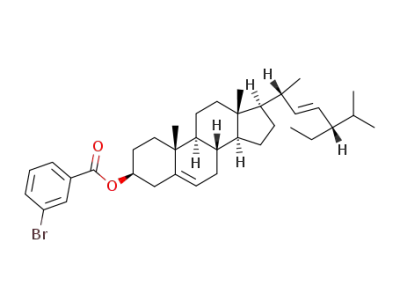
3β-(3-bromo-benzoyloxy)-stigmasta-5,22t-diene
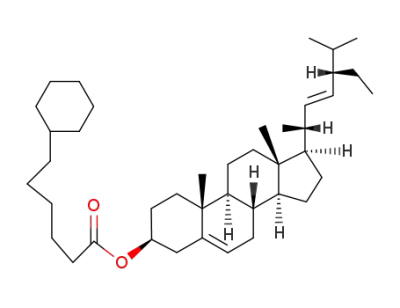
3β-(6-cyclohexyl-hexanoyloxy)-stigmasta-5,22t-diene
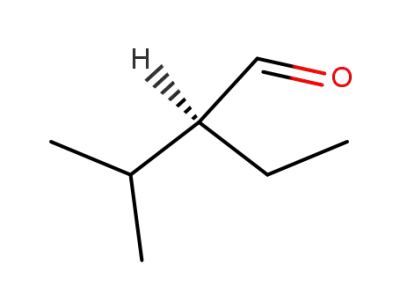
(S)-2-ethyl-3-methylbutanal
β-stigmasterol

