Current location:Home > Markets > Pharmaceuticals


|
Biochem/physiol Actions |
D-(?)-Salicin exerts anti-rheumatism and anti-inflammatory activities. It inhibits lipopolysaccharide (LPS) induced inflammation in in vitro and in vivo studies. It regulates different signaling pathways such as nuclear factor kappa B (NF-κB) and mitogen-activated protein kinase (MAPK). It also regulates cytokines concentration such as tumor necrosis factor-alpha(TNF-α), interleukin-1β, interleukin-6, and interleukin-10. |
|
Purification Methods |
Crystallise D(-)-salicin from EtOAc, EtOH or water and sublime it at 190-195o/12mm. [Armour et al. J Chem Soc 412 1961, IR: Pearl & Darling J Org Chem 24 731 1959, Beilstein 17 III/IV 2986, 17/7 V 113.] |
|
Definition |
ChEBI: An aryl beta-D-glucoside that is the beta-D-glucoside of the phenolic hydroxy group of salicyl alcohol. |
|
General Description |
Salicin is a non-phenolic glucosidic compound extracted from meadowsweet (Filipendula ulmaria L). It is majorly used as a substitute for quinine. Salicin can be used as a therapeutic for patients suffering from rheumatic fever. Salicin acts a metabolic precursor for salicylic acid. It is a novel phytochemical that exhibits immunological cross functions in plants and humans. Salicin facilitates growth and reproduction in plants. In addition, It also protects plants against biotic and abiotic stress. |
InChI:InChI=1/C13H18O7/c14-5-7-3-1-2-4-8(7)19-13-12(18)11(17)10(16)9(6-15)20-13/h1-4,9-18H,5-6H2/t9-,10-,11+,12-,13-/m0/s1
The chemical constituents of Homalium co...
In the present work, the synthesis of ac...
Adopting the ‘remote activation concept’...
The stereoselective xylosylation of (RS)...
The glucosylation of some coumarin and f...
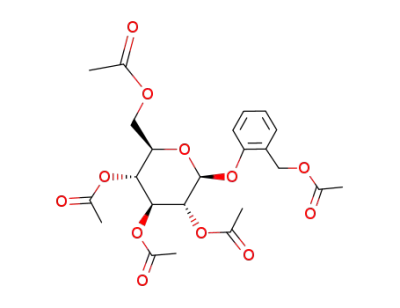
salicin pentaacetate

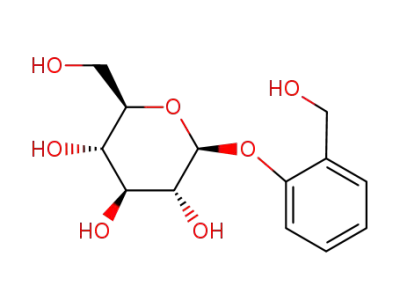
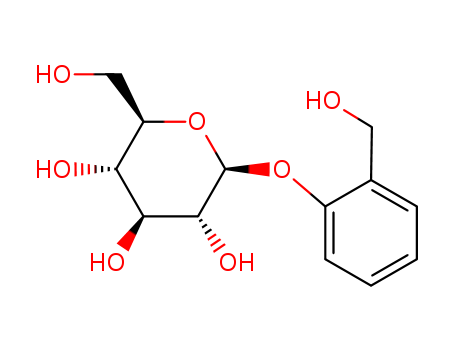
D-(-)-salicin
| Conditions | Yield |
|---|---|
|
With
sodium methylate;
In
methanol;
at 20 ℃;
for 1h;
Inert atmosphere;
|
80 mg |
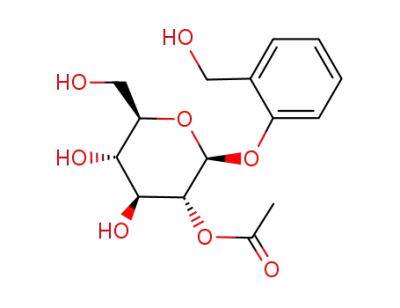
2-(hydroxymethyl)phenyl-2-O-acetyl-β-D-glucopyranoside


D-(-)-salicin
| Conditions | Yield |
|---|---|
|
With
sodium methylate;
In
methanol;
at 20 ℃;
for 2h;
|
99% |
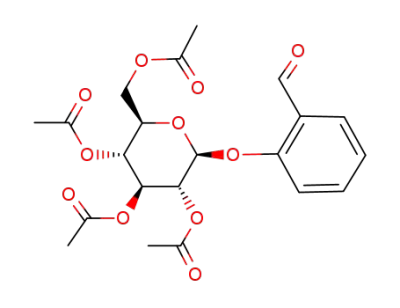
helicin acetate
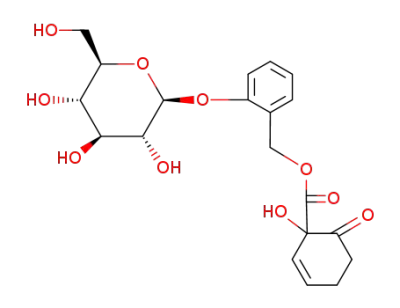
2-[[[(1-hydroxy-6-oxo-2-cyclohexen-1-yl)carbonyl]hydroxy]methyl]phenyl-β-D-glucopyranoside
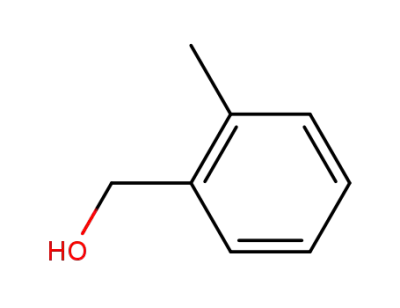
2-methyl-benzyl alcohol
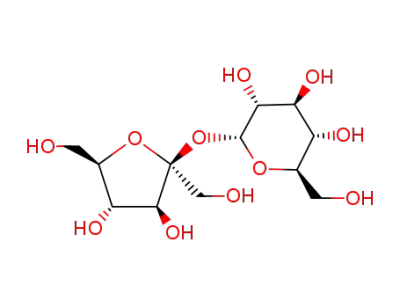
Sucrose
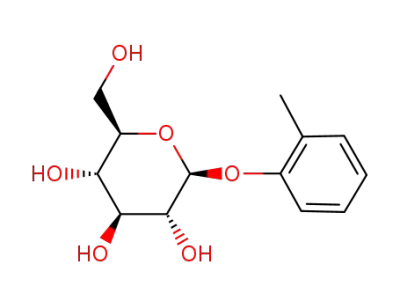
o-tolyl-β-D-glucopyranoside
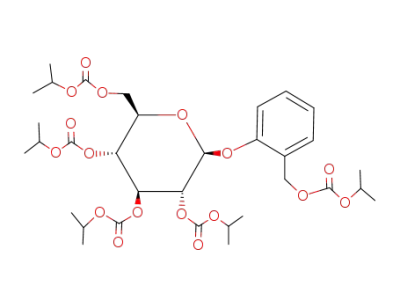
Penta-O-(isopropoxycarbonyl)salicin
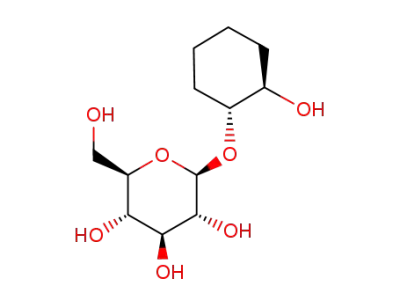
(1R,2R)-trans-1,2-cyclohexanediol-1-O-β-D-glucopyranoside
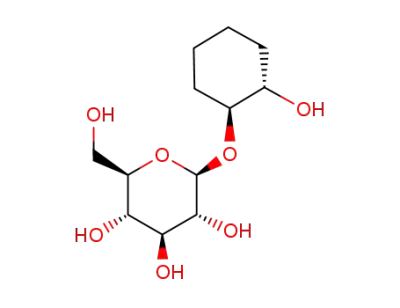
(1S,2S)-trans-1,2-cyclohexanediol-1-O-β-D-glucopyranoside

