

|
source |
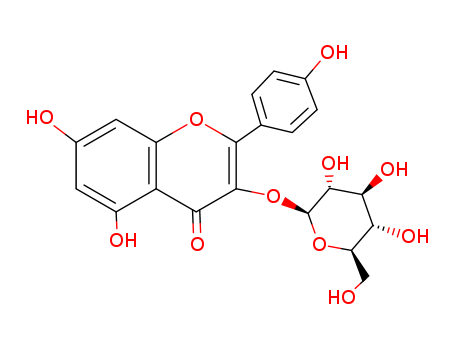
Astragalin belongs to the class of organic compounds known as flavonoid-3-o-glycosides. These are phenolic compounds containing a flavonoid moiety which is O-glycosidically linked to carbohydrate moiety at the C3-position. Astragalin exists as a solid, slightly soluble (in water), and a very weakly acidic compound (based on its pKa). Within the cell, astragalin is primarily located in the cytoplasm. Astragalin can be converted into astragalin heptaacetate and 2''-acetylastragalin. Outside of the human body, astragalin can be found in a number of food items such as tamarind, american cranberry, chickpea, and bilberry. This makes astragalin a potential biomarker for the consumption of these food products. |
|
Overview |
Astragalin is a chemical compound. It can be isolated from Phytolacca americana (the American pokeweed) or in the methanolic extract of fronds of the fern Phegopteris connectilis.t is also found in wine. |
|
Definition |
ChEBI: A kaempferol O-glucoside in which a glucosyl residue is attached at position 3 of kaempferol via a beta-glycosidic linkage. |
InChI:InChI=1/C21H20O11/c22-7-13-15(26)17(28)18(29)21(31-13)32-20-16(27)14-11(25)5-10(24)6-12(14)30-19(20)8-1-3-9(23)4-2-8/h1-6,13,15,17-18,21-26,28-29H,7H2/t13-,15-,17+,18-,21+/m1/s1
From the aerial parts of Phlomis spinide...
-
Key Word Index - Picea abies; Pinaceae; ...
The glycosylation of small hydrophobic c...
Engineering the function of triterpene g...
A highly regio-specific and donor-promis...
Icariin (1a), a 7-O-glycosylated flavono...
kaempferol 3-O-[2'',6''-di-O-(trans-p-coumaroyl)]-β-D-glucopyranoside

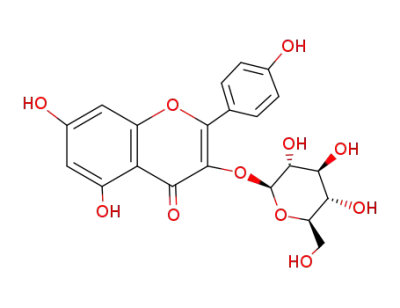
astragalin

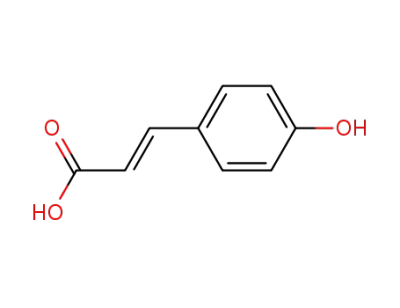
p-Coumaric Acid
| Conditions | Yield |
|---|---|
|
With
potassium hydroxide;
In
1,4-dioxane;
for 3h;
|
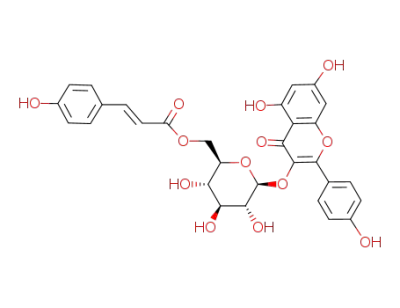
trans-tiliroside


astragalin


p-Coumaric Acid
| Conditions | Yield |
|---|---|
|
With
sodium hydroxide;
for 2.5h;
Product distribution;
Ambient temperature;
hydrolysis;
|
10 mg |
|
With
sodium hydroxide;
at 50 - 60 ℃;
|
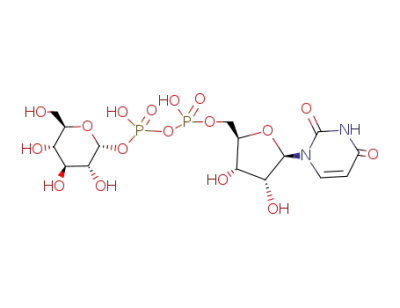
UDP-glucose
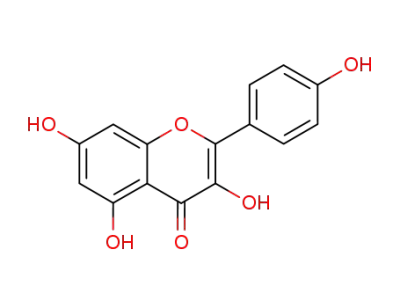
kaempferol
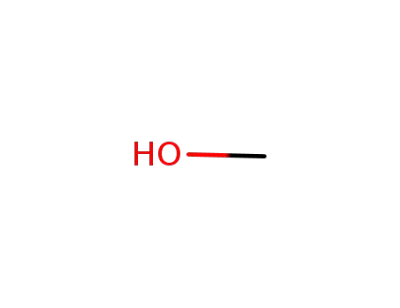
methanol
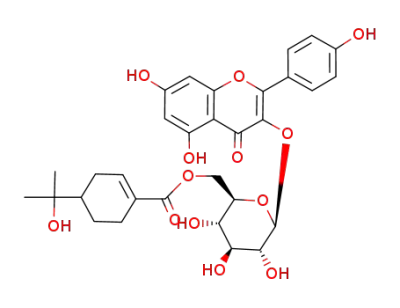
resinoside A

kaempferol
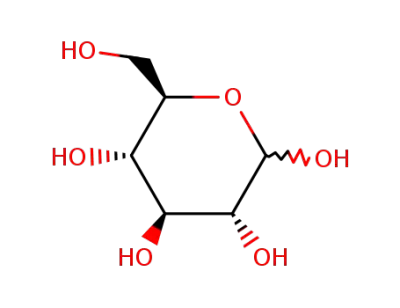
D-Glucose
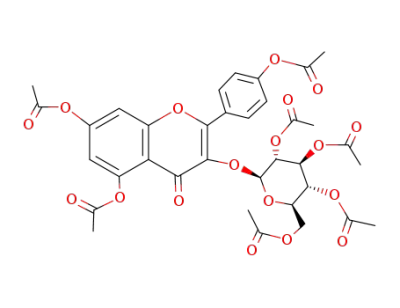
Astragalin-acetat
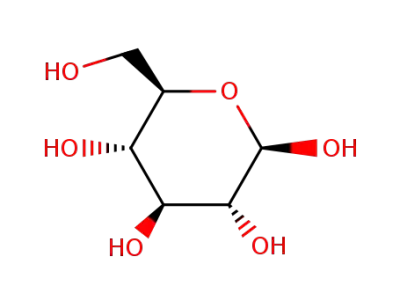
β-D-glucose

