Current location:Home > Markets > Nutraceuticals


|
Pharmacological action |
Chlorogenic acid is also called "green" auspicious acid, tannins "coffee", "coffee tannic acid", is a plant produce a styrene acrylic compounds through shikimic acid hitches in aerobic respiration process.It is coffee acyl quinic acid derivatives.The oxidation resistance is strong, and also has the anti HIV, anti tumor cells, antibacterial, improve central excited, cholagogue function.It has teratogenic, allergy and regulate the activity of cytochrome P450 ligase, and other functions.It is widely exist in more content honeysuckle plants. It is obtained by the water needle crystal, and it is the hemihydrate, when 110 ℃ it is to hydrate. Melting point is 208 ℃, [a] D25 33.5 ° (water, C = 1), K = 2.2 x 10-3 (27 ℃).It is weak acidity and convergence. When 25 ℃ , the solubility in water is 4%, easily soluble in hot water, but soluble in ethanol, acetone, slightly soluble in ethyl acetate, insoluble in chloroform, ether and carbon disulfide. It is azeotropic with concentrated hydrochloric acid and come to blue dye. It can make the bromine water fade, It is azeotropic with strong hydrogen iodide and obtain benzoic acid, It is obtained phenol by thermal decomposition together with water at 230 ℃, It decomposite to caffeic acid and quinic acid with dilute hydrochloric acid, and caffeic acid and quinic acid with dilute alkali at room temperature. Chlorogenic acid and its isomer different chlorogenic acid and green chlorogenic acid are in fruits of dicotyledonous plants, leaves, and other organizations,which is an important factor for plant metabolism. Chlorogenic acid has good biological activity, the transparent acid enzyme and inhibition of glucose-6-phosphatase, free radical lipid perxidation qing mutagenesis resistance and cardiovascular crown reinforcement, special efficacy in lipid-lowering, both anti-bacterial anti-virus, protecting liver cholagogic. Chlorogenic acid as the condensation product of caffeic acid and quinic acid, is blue when meeting with iron, which is similar to tannins , but not precipitated with protein, so no convergence. Caffeic acid and chlorogenic acid has the wide bacteriostasis, but can be inactivated protein in the body. This product is similar to caffeine, oral or intraperitoneal injection, can improve the rat central excitability. Oral the drug of chlorogenic acid and coffee acid can increase the amount of hydrochloric acid secretion in the stomach, and can make slow pulse, and quinic acid is not. Chlorogenic acid could significantly increase small intestine peristalsis on the rat or mouse. Chlorogenic acid, caffeic acid and quinic acid can increase the tension of rat uterus, this function can be cancelled by papaverine, while atropine can't influence. Caffeic acid, chlorogenic acid, also can promote bile secretion in rats. On the specimens of rabbit ileum in vitro, this product can enhance the effect of adrenaline, but has no effect on blood glucose of adrenaline. |
|
Protecting liver, gallbladder |
Chlorogenic acid in flos lonicerae has favorable gallbladder function, promoting biliary Secretion of rats. Injection of 200 mg/kg chlorogenic acid to browns MAO honeysuckle total saponins subcutaneous can be significantly against CCl4, paracetamol and the increase of serum GPT caused by galactosamine in mice liver and liver triglyceride levels, and significantly reduce the severity of the liver pathological damage, making dot necrosis number sum and necrosis occur rate liver decreased obviously. |
|
The inhibitory effect on hyaluronidase and glucose - 6 - phosphatase |
Hyaluronic cranberry (HAase) is one of the cracking mucopolysaccharide enzymes, can catalyze hyaluronic acid (HA) decomposition,which is in relation to the permeability of vascular system and inflammation. HA is a mucopolysaccharide, which is composed of uronic acid and acetyl glucosamine, having a variety of functions, such as heal, make the skin wetting health, lubricating joints and prevent inflammation, etc. 3, 5-2 coffee acyl quinic acid (artichoke acid) and chlorogenic acid found in the ethyl acetate extract from the narrow assists cone flower (Echinacea amgustifolia DC) roots have stronger inhibitory effect of HAase activated. Animal in vitro studies have demonstrated that chlorogenic acid can not reversible inhibition of glucose-6-hydrolysis of phosphatase, reduce liver glycogen decomposition and exogenous glucose absorption. Animal studies have shown that the use of chlorogenic acid can reduce high blood sugar spikes caused by the use of glucagon (glycogenolysis). Therefore, chlorogenic acid can lower blood sugar level, improve the liver glucose-6-phosphoric acid and the concentration of hepatic glycogen. |
|
The character of molecular structure |
Chlorogenic acid is formed by caffeic acid and quinic acid, and the molecular structure has ester bond, unsaturated double bond and polyphenol unstable part of the three. In the process of extracted from plants, chlorogenic acid often happen by hydrolysis and intramolecular ester base migration isomerization . Due to the special structure of chlorogenic acid, it can extract from plants by the polarity solvent such as ethanol, acetone, methanol. But due to the instability of chlorogenic acid itself, extraction can not be in high temperature, strong light and heat for a long time. Chlorogenic acid liquid were placed in brown bottle, refrigerator (2 ℃) to save to be the most stable. |
|
Preparation method |
It can be obtained by (1S)-1-ethoxyformyloxy-3-cis-[3,4-dihydroxy-anti-cinnamoyl oxygen radicals]-4'-anti-5'-anti-dihydroxy cyclohexane-γ-methyl formate with barium hydroxide in methanol catalytic hydrolysis, or by the segregation from green coffee beans. |
|
HPLC method for determination the content |
(1)Chromatographic conditions: octadecyl silane bonded silica gel as the filling agent; Acetonitrile-0.4% phosphoric acid solution (13:87) as mobile phase; Detection wavelength of 327 nm. Theoretical plate number according to the chromatographic peak of chlorogenic acid is greater than 1000. (2)the preparation of the reference substance solution: precisely weigh the right amount of chlorogenic acid reference substance, placed in brown volumetric flask, adding 50% methanol solution containing 40 μg /mL (saving below 10 ℃). (3)the preparation of sample solution: taking samples of dry powder is about 0.5 g, precision said, with a plug in the conical flask, adding 50 ml 50% methanol , 30 min ultrasonic processing , cooling, weighing, make up the weight loss reduction with 50% methanol, shake well, filtration, abandoned the early filtrate, precision measuring duration of filtrate 5 ml, 25 ml volumetric brown flask , add 50% methanol to the scale, shake well. (4)assimilate precision respectively from each 10 μL reference substance and the sample solution, injection liquid chromatograph, determination,obtained. |
|
Toxicity |
Chlorogenic acid is sensitive to people, can cause asthma, dermatitis, but no this reaction to the oral of chlorogenic acid. It can be active no sensitization substances by small intestinal secretions convertion to. The toxicity of chlorogenic acid is very small, LD50 is greater than 1 g/kg to young rats, intraperitoneal injection of greater than 0.25 g/kg. |
|
Chemical property |
Hemihydrate is needle crystal (water). When 110 ℃, it become to anhydrous compounds. Melting point: 208 ℃, [alpha] 20D-35.2 (C = 2.8). When 25 ℃, water solubility was 4%, solubility is bigger in the hot water. Soluble in ethanol and acetone, slightly soluble in ethyl acetate. |
|
Toxicity grading |
toxication |
|
Acute toxicity |
Abdominal cavity-rat LDL0:4000 mg/kg |
|
Flammability hazard characteristics |
Combustible; Release acrid smoke fire |
|
Storage and transport properties |
Low temperature warehouse, ventilated, dry |
|
Fire extinguishing agent |
Water, carbon dioxide, dry powder, sand |
|
Purification Methods |
Crystallise the acid from water and dry it at 110o. [Beilstein 10 H 537, 10 I 271, 10 II 378, 10 III 2408, 10 IV 2259.] |
|
Chemical Composition and Properties |
Chlorogenic acid (CGA) is a depside acid produced by caffeic acid and quinic acid. CGA is a phenylacrylate polyphenol compound synthesized in plants during aerobic respiration. It exists in various isomeric forms, including mono-caffeoyl quinic acid and dicaffeoyl quinic acid. |
|
Sources |
Found in a wide range of plants, fruits, vegetables, and beverages, with coffee being a prominent source. Content varies among sources, with high levels in green coffee beans, prunes, apple, tomato, eggplant, carrot, tea, and herbal teas like jasmine and green tea. |
|
Biological Activities |
CGA exhibits various important biological activities, making it valuable in medicine, food, and other industries. |
|
Metabolism and Absorption |
In rats, CGA is absorbed in the stomach and further hydrolyzed to caffeic acid and quinic acid in the intestine. After absorption, CGA is metabolized into glucuronic acid glycosides and sulfate metabolites. CGA's oral absorption rate is low, and it mainly exists as metabolites in plasma before excretion through the kidneys. |
|
Biotechnological Significance |
Understanding CGA biosynthesis pathways and enhancing its content in medicinal plants and fungi through biotechnology is of great significance. With its diverse biological activities, CGA is considered internationally as a valuable bioactive substance with potential applications in various fields. |
|
General Description |
Chlorogenic acid, a hydroxycinnamoyl quinate ester, plays a role in the storage of tropane alkaloids such as cocaine in *Erythroxylum coca* by forming physical complexes with these alkaloids, as demonstrated through spectroscopic analysis. Its biosynthesis involves a BAHD acyltransferase enzyme, and its accumulation correlates with tropane alkaloid levels, suggesting its function as a stabilizing or storage partner for these compounds in the plant. |
|
Category |
Toxic substances |
InChI:InChI=1/C16H18O9/c17-9-3-1-8(5-10(9)18)2-4-13(20)25-12-7-16(24,15(22)23)6-11(19)14(12)21/h1-5,11-12,14,17-19,21,24H,6-7H2,(H,22,23)/b4-2+/t11-,12-,14-,16+/m1/s1
Eleven new diglycosides, erycibosides A-...
An efficient and versatile synthesis of ...
Phenolic compounds of sainfoin (Onobrych...
Polyphenols are secondary plant compound...
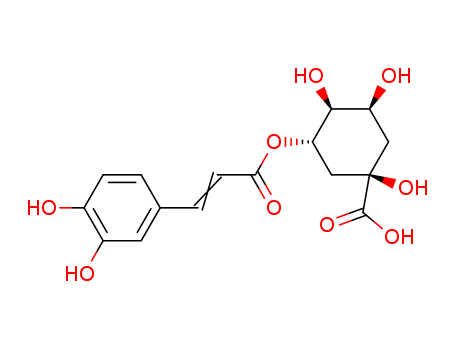
neochlorogenic acid

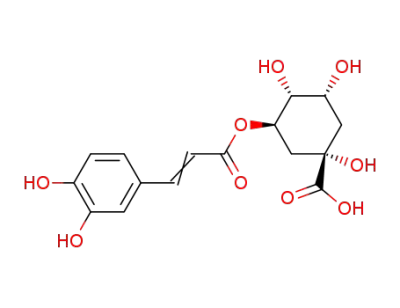
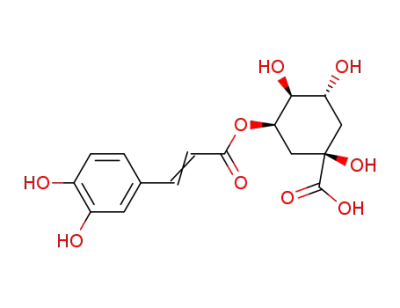
chlorogenic acid

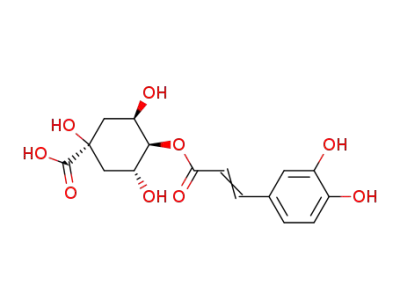
4-O-caffeoylquinic acid
| Conditions | Yield |
|---|---|
|
In
water;
at 100 ℃;
for 0.5h;
pH=8;
|
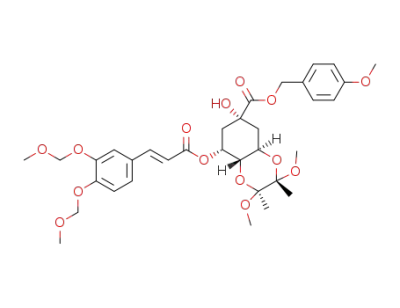
4-methoxybenzyl (2'S,3'S)-3-O,4-O-(2',3'-dimethoxybutane-2',3'-diyl)-5-O-(3,4-di-O-methoxymethylcaffeoyl)quinate

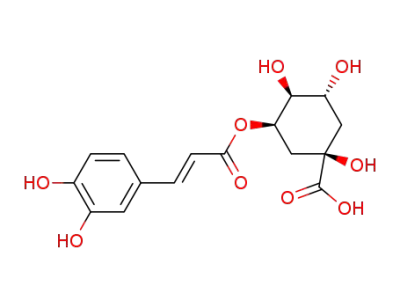
neochlorogenic acid
| Conditions | Yield |
|---|---|
|
With
pentamethylbenzene,; boron trichloride;
In
dichloromethane;
at -40 - 0 ℃;
for 3h;
Reagent/catalyst;
Inert atmosphere;
|
69% |

neochlorogenic acid
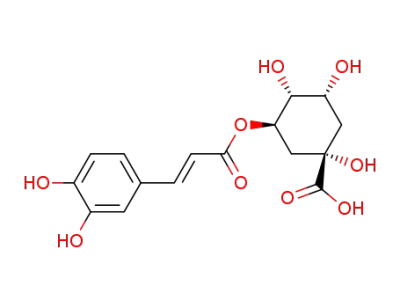
3-caffeoylquinic acid
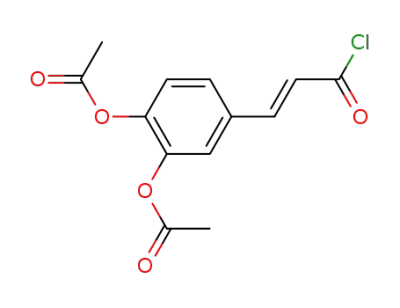
(E)-3,4-di-O-acetylcaffeoyl chloride
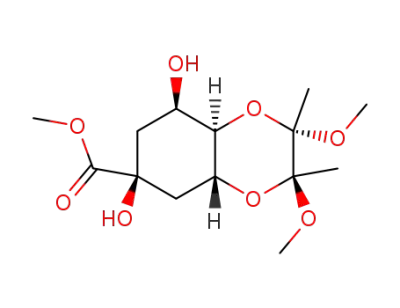
(2S,3S,4aR,6S,8R,8aR)-6,8-Dihydroxy-2,3-dimethoxy-2,3-dimethyl-octahydro-benzo[1,4]dioxine-6-carboxylic acid methyl ester
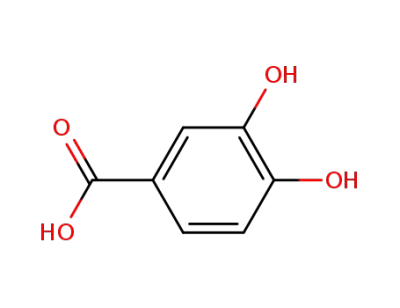
3,4-Dihydroxybenzoic acid
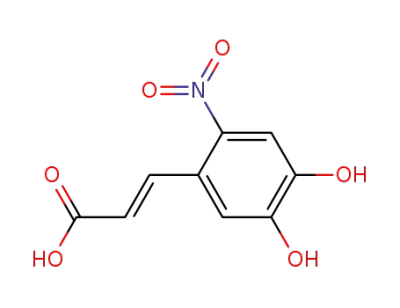
(E)-3-(4,5-dihydroxy-2-nitrophenyl)propenoic acid
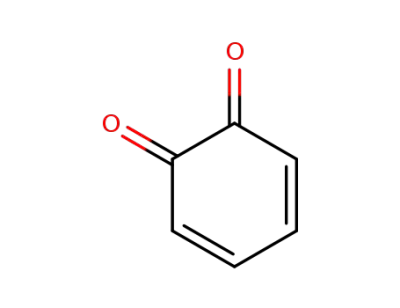
ortoquinone
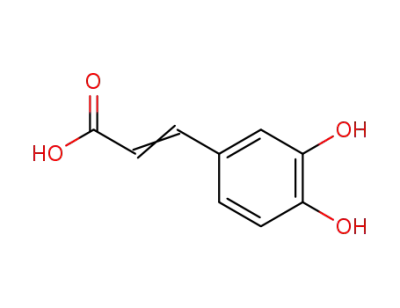
caffeic acid

