Current location:Home > Markets > Pharmaceuticals


|
History |
In 1911, Gorter firstly isolated a crystalline substance from Andrographis paniculata, identified it as a diterpene lactone compound, and named it andrographolide .Andrographolide structure is complex and difficult to artificially synthesize, so it is often extracted from the plant.Andrographolide water solubility is poor and its bioavailability is low; its pharmacological effects are extensive but weak, so the preparation requirements are strict. Therefore, from the 1970s, drugs and organic chemical researchers have done a lot of work in the modification and transformation of andrographolide, mainly concentrated in the α, β-unsaturated lactone double bond Michael addition, redox, selective esterification of hydroxyl groups, oxidation and substitution reactions, intramolecular cyclization, replacement of lactone rings, etc. They had obtained a number of new derivatives. In the research of pharmacological effects, the main study of the andrographolide derivatives focuses on antitumor, antivirus, and other biological activities and has made some progress . |
|
Biochem/physiol Actions |
Diterpenoid lactone with anti-inflammatory properties. It blocks T-cell proliferation to allogenic stimuli and the chemotactic migration of macrophages induced by complement. Blocks the proliferation of several cancer cell lines in vitro. |
|
Pharmacology |
Modern pharmacological studies have shown that andrographolide has effects of anti-inflammatory, antibacterial, antivirus, antitumor, and immune regulation and can be used in treatment of cardiovascular-cerebrovascular diseases and protectionof the liver and gallbladder. Andrographolide can treat the fever caused by pneumococcus and hemolytic Streptococcus mutans, mainly by inhibiting the hypothalamus PGE2 and cAMP content to exert its antipyretic, nonetheless reducing the chemotaxis (fMLP) CD11b + and CD18 + may be the main mechanism of its antiinflammatory effect. Andrographolide has antagonistic effects on Hong Kong virus (HKV), Ebola virus (EBOV), and respiratory syncytial virus (RSV) and has been shown to inhibit HIV, SARS, and viral myocarditis ST2 in?vitroAndrographolide can also ameliorate myocardial ischemia and serve as protection from ischemia-reperfusion injury. It also has protection effect of vascular endothelial cells as well as can regulate lipid disorder, lower blood pressure, and exert effect of anti-atherosclerosis. It can also prevent angiogenesis after restenosis and improve blood rheology. Its mechanisms are mainly due to its free radical scavenging and antiplatelet aggregation properties. Andrographolide can increase the bile flow, bile salt, bile acid, and deoxycholic acid in experimental rats and guinea pigs and can reverse the reduction of bile and cholic acid and other secretions caused by paracetamol and improve liver function . |
|
Anticancer Research |
It is a labdane diterpenoid, which shows cytotoxic effect against different cancercell lines like human epidermoid cancer cells (KB), lymphocytic leukemia cells(P388), breast cancer cells (MCF-7), and colon cancer cells (HCT-116). It inhibitsthe proliferation of colon cancer cells (HT-29) and exerts pro-differentiative effecton mouse myeloid leukemia M1 cell line (Desai et al. 2008). |
|
in vitro |
andrographolide was found to suppress the expression of inducible nitric oxide synthase in a concentration-dependent manner. in the andrographolide-treated group, a reduction of akt, c-jun n-terminal kinase and p65 phosphorylation was observed. andrographolide also caused a decrease in bcl-2/nf-κb expression and a dose-dependent increase in the expression of cleaved-caspase3/bax protein [1]. |
|
in vivo |
rats were dosed with andrographolide intragastrically for 5 consecutive days in a hepatoprotection study. results indicated that andrographolide could up-regulate glutamate cysteine ligase catalytic and modifier subunits, heme oxygenase-1, superoxide dismutase-1, glutathione s-transferase protein and mrna expression in the heart, liver, and kidney [2]. |
|
IC 50 |
11.0 ± 0.28 μg/ml for α-glucosidase; 11.3 ± 0.29 μg/ml for α-amylase |
|
Definition |
ChEBI: Andrographolide is a labdane diterpenoid isolated from the leaves and roots of Andrographis paniculata that exhibits anti-HIV, anti-inflammatory and antineoplastic properties. It has a role as a metabolite, an anti-inflammatory drug, an anti-HIV agent and an antineoplastic agent. It is a gamma-lactone, a primary alcohol, a secondary alcohol, a labdane diterpenoid and a carbobicyclic compound. |
|
Application |
Andrographolide may be used as a reference standard for the determination of andrographolide in:Leaves of Andrographis paniculata by soxhlet extraction-recrystallization-column chromatography purification followed by analyses using reversed phase high performance liquid chromatography (RP-HPLC) as well as gas chromatography-mass spectrometry (GC-MS).Roots of Andrographis paniculata by HPLC with diode array detection (DAD), gas chromatography-mass spectrometry (GC-MS) and LC coupled to tandem mass spectrometry (MS/MS) equipped with electrospray ionization (ESI) and multiple reaction monitoring (MRM) mode of detection.Stems of Andrographis paniculata by ultra-HPLC combined coupled to triple quadrupole-linear ion trap mass spectrometry (QqQLIT-ESI-MS/MS) with MRM detection.Rat plasma samples by protein precipitation and subsequent ESI-LC-MS/MS determination using MRM detection. |
|
General Description |
Andrographolide, a bioactive diterpene lactone, is the main constituent of Andrographis paniculata, a plant used in traditional medicines. It is known to have different biological activities such as anti-inflammatory, anti-malarial, anti-cancer and hepatoprotective activity. It also shows potent anti-viral effect against dengue virus. |
InChI:InChI=1/C20H30O5/c1-12-4-7-16-19(2,9-8-17(23)20(16,3)11-21)14(12)6-5-13-15(22)10-25-18(13)24/h5,14-17,21-23H,1,4,6-11H2,2-3H3/b13-5+/t14-,15-,16?,17-,19+,20+/m1/s1
An enantioselective total synthesis of t...
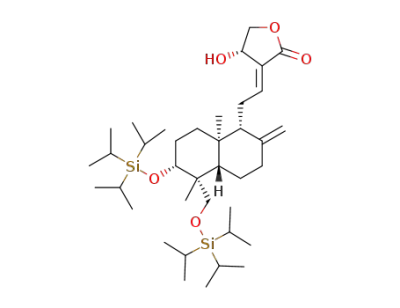
(S,E)-3-(2-((1R,4aS,5R,6R,8aS)-5,8a-dimethyl-2-methylene-6-((triisopropylsilyl)oxy)-5-(((triisopropylsilyl)oxy)methyl)decahydronaphthalen-1-yl)ethylidene)-4-hydroxydihydrofuran-2(3H)-one

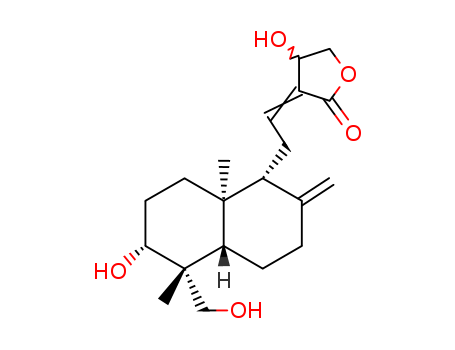
andrographolide
| Conditions | Yield |
|---|---|
|
With
pyridine; pyridine hydrogenfluoride;
In
tetrahydrofuran;
at 0 - 20 ℃;
for 34h;
Inert atmosphere;
|
92% |

((4aS,5R,6R,8aR)-5,8a-dimethyl-6-((triisopropylsilyl)oxy)-5-(((triisopropylsilyl)oxy)methyl)-3,4,4a,5,6,7,8,8a-octahydronaphthalene-1,2-diyl)dimethanol


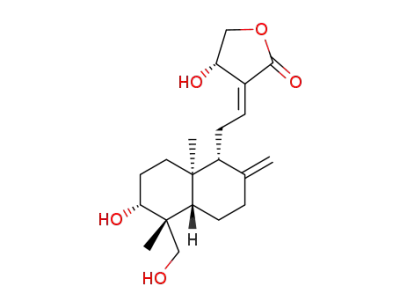
andrographolide
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 5 steps
1.1: di-isopropyl azodicarboxylate; triphenylphosphine; 2-nitrobenzenesulfonyl hydrazide / tetrahydrofuran / 7.5 h / -30 - 25 °C / Inert atmosphere
2.1: triphenylphosphine; 1H-imidazole; iodine / tetrahydrofuran / 24 h / 0 - 20 °C / Inert atmosphere
3.1: tert.-butyl lithium / diethyl ether; pentane / 1 h / -78 °C / Inert atmosphere
3.2: 0.08 h / -78 - 0 °C / Inert atmosphere
4.1: triethylamine; palladium diacetate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; sodium carbonate; tetra-(n-butyl)ammonium iodide / 1,4-dioxane / 15 h / 80 °C / Inert atmosphere; Schlenk technique
5.1: pyridine hydrogenfluoride; pyridine / tetrahydrofuran / 34 h / 0 - 20 °C / Inert atmosphere
With
pyridine; 1H-imidazole; di-isopropyl azodicarboxylate; 2-nitrobenzenesulfonyl hydrazide; iodine; tert.-butyl lithium; palladium diacetate; tetra-(n-butyl)ammonium iodide; sodium carbonate; pyridine hydrogenfluoride; triethylamine; triphenylphosphine; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene;
In
tetrahydrofuran; 1,4-dioxane; diethyl ether; pentane;
|

geraniol
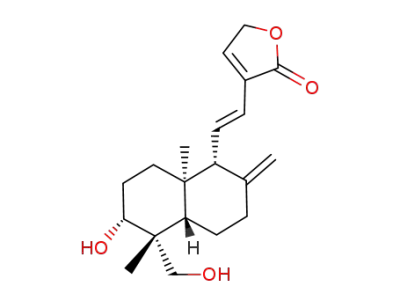
14-deoxy-11,12-didehydroandrographolide
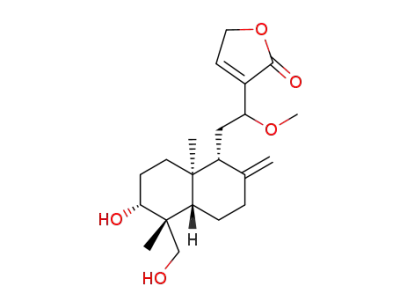
14-deoxy-12-methoxy-andrographolide
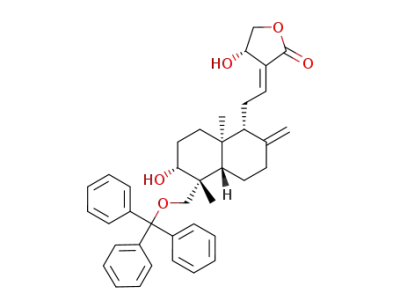
19-Tr-andrographolide
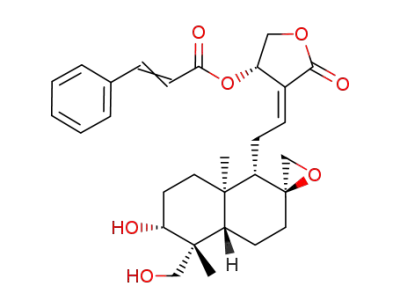
C29H36O7

