

Quality products make an important contribution to long-term revenue and profitability. Quality manufacturer supply Idebenone 58186-27-9 in stock with high standard
idebenone is an anti-oxidant capable of protecting the skin from a variety of free-radical attacks, including the formation of secondary chemicals that negatively affect skin physiology. It is said to improve intrinsic as well as extrinsic skin damage caused by freeradical formation. Idebenone is a synthetically manufactured form of coenzyme Q10 and has a smaller molecular structure. This allows it to penetrate the skin and apparently the cellular membrane. Clinical studies demonstrate a visible improvement in photodamaged skin, reduced skin roughness and dryness, decreased fine lines and wrinkles, and increased skin hydration. In addition, idebenone helps improve hyperpigmentation because its molecular structure is similar to that of hydroquinone. Although most of the associated benefits are seen primarily in the epidermis, some increase in dermal collagen has also been confirmed. Idebenone has been used for such health-related problems as Alzheimer’s and heart disease.

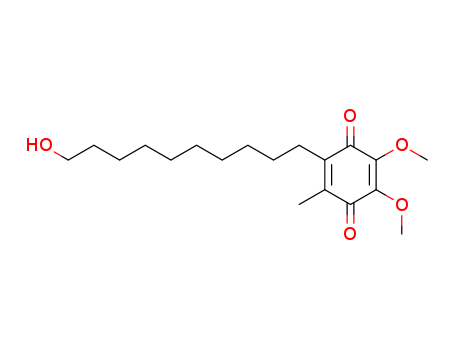
Idebenone

Sucrose
| Conditions | Yield |
|---|---|
|
|
9-bromononan-1-ol


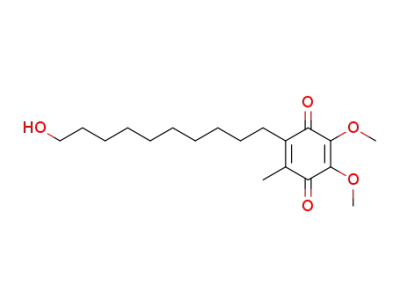
Idebenone
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 9 steps
2: 95 percent / dimethylsulfoxide / 15 h / 80 °C
3: 86 percent / NaOH, 3N HCl / ethanol / 19 h / Heating
4: PCl5 / CH2Cl2 / 2 h / 25 °C
5: 44 percent / AlCl3 / 1,2-dichloro-ethane / 72 h / 20 °C
6: 79 percent / NaOH / methanol / 2 h / Ambient temperature
7: H2, 70percent HClO4 / 5percent Pd-C / 24 h / Ambient temperature
8: NaOH / methanol / 24 h / 5 °C
9: O2 / salcomine / dimethylformamide / Ambient temperature; reaction time, additive
With
hydrogenchloride; sodium hydroxide; aluminium trichloride; perchloric acid; phosphorus pentachloride; hydrogen; oxygen;
palladium on activated charcoal; salcomine;
In
methanol; ethanol; dichloromethane; dimethyl sulfoxide; 1,2-dichloro-ethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 9 steps
2: 95 percent / dimethylsulfoxide / 15 h / 80 °C
3: 86 percent / NaOH, 3N HCl / ethanol / 19 h / Heating
4: PCl5 / CH2Cl2 / 2 h / 25 °C
5: 44 percent / AlCl3 / 1,2-dichloro-ethane / 72 h / 20 °C
6: 79 percent / NaOH / methanol / 2 h / Ambient temperature
7: H2, 70percent HClO4 / 5percent Pd-C / 24 h / Ambient temperature
8: NaOH / methanol / 24 h / 5 °C
9: O2 / salcomine / dimethylformamide / Ambient temperature; reaction time, additive
With
hydrogenchloride; sodium hydroxide; aluminium trichloride; perchloric acid; phosphorus pentachloride; hydrogen; oxygen;
palladium on activated charcoal; salcomine;
In
methanol; ethanol; dichloromethane; dimethyl sulfoxide; 1,2-dichloro-ethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 9 steps
2: 95 percent / dimethylsulfoxide / 15 h / 80 °C
3: 86 percent / NaOH, 3N HCl / ethanol / 19 h / Heating
4: PCl5 / CH2Cl2 / 2 h / 25 °C
5: AlCl3 / 1,2-dichloro-ethane / 5 degC, 2 h; 20 degC, 70 h
6: 79 percent / NaOH / methanol / 2 h / Ambient temperature
7: H2, 70percent HClO4 / 5percent Pd-C / 24 h / Ambient temperature
8: NaOH / methanol / 24 h / 5 °C
9: O2 / salcomine / dimethylformamide / Ambient temperature; reaction time, additive
With
hydrogenchloride; sodium hydroxide; aluminium trichloride; perchloric acid; phosphorus pentachloride; hydrogen; oxygen;
palladium on activated charcoal; salcomine;
In
methanol; ethanol; dichloromethane; dimethyl sulfoxide; 1,2-dichloro-ethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 9 steps
2: 95 percent / dimethylsulfoxide / 15 h / 80 °C
3: 86 percent / NaOH, 3N HCl / ethanol / 19 h / Heating
4: PCl5 / CH2Cl2 / 2 h / 25 °C
5: 29 percent / AlCl3 / 1,2-dichloro-ethane / 72 h / 20 °C
6: 79 percent / NaOH / methanol / 2 h / Ambient temperature
7: H2, 70percent HClO4 / 5percent Pd-C / 24 h / Ambient temperature
8: NaOH / methanol / 24 h / 5 °C
9: O2 / salcomine / dimethylformamide / Ambient temperature; reaction time, additive
With
hydrogenchloride; sodium hydroxide; aluminium trichloride; perchloric acid; phosphorus pentachloride; hydrogen; oxygen;
palladium on activated charcoal; salcomine;
In
methanol; ethanol; dichloromethane; dimethyl sulfoxide; 1,2-dichloro-ethane; N,N-dimethyl-formamide;
|
The CAS number of Idebenone is 58186-27-9.
More information of Idebenone 58186-27-9 are:
|
CAS Number |
58186-27-9 |
|
Density |
1.08 g/cm3 |
|
Melting Point |
52-55 °C |
|
Boiling Point |
497.3 °C at 760 mmHg |
|
Flash Point |
170.1 °C |
|
Vapor Pressure |
5.67E-12mmHg at 25°C |
|
Refractive Index |
1.502 |
|
HS CODE |
2933399990 |
|
PSA |
72.83000 |
|
LogP |
3.46230 |
|
Pka |
15.20±0.10(Predicted) |
Synonyms for Idebenone 58186-27-9:2,5-Cyclohexadiene-1,4-dione,2-(10-hydroxydecyl)- 5,6-dimethoxy-3-methyl-;Idebenone (JAN);2,5-Cyclohexadiene-1,4-dione, 5,6-dimethoxy-2-(10-hydroxydecyl)-3-methyl-;6-(10-Hydroxydecyl)-2,3-dimethoxy-5-methyl-1,4-benzoquinone;CV 2619;Idebenone [GMP];2-(10-Hydroxydecyl)-5,6-dimethoxy-3-methyl-p-benzoquinone;Idebenonum [Latin];Idebenone [INN:JAN];Idebenona [GMP];Idebenona [Spanish];2-(10-hydroxydecyl)-5,6-dimethoxy-3-methyl-cyclohexa-2,5-diene-1,4-dione;Idenbenone;
The chemical formula of Idebenone is C19H30O5 which containing 19 Carbon atoms,30 Hydrogen atoms and 5 Oxygen atoms,and the molecular weight of Idebenone is 338.444.
Idebenone is an organic compound belonging to the quinone family, being similar to coenzyme Q-10. It is a kind of drug developed by Takeda Pharmaceutical Company for the treatment of Alzheimer’s disease and some other cognitive defects. However, these have been not very much progress associated with this indication. It is now also used for the treatment of Friedreich’s ataxia with a positive effect on cardiac hypertrophy and neurological function. However, this indication is only approved in Canada, not in Europe and US. It is now under investigation on its efficacy for the treatment of Duchenne muscular dystrophy, Leber’s hereditary optic neuropathy, mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes) as well as primary progressive multiple sclerosis. The efficacy of this drug still demands more evidences.
InChI:InChI=1/C19H30O5/c1-14-15(12-10-8-6-4-5-7-9-11-13-20)17(22)19(24-3)18(23-2)16(14)21/h20H,4-13H2,1-3H3
Relevant articles related to Idebenone:
|
Article |
Source |
|
A Simple and Convenient Two-step Synthesis of Idebenone |
Zhou, Rong-Ye,Li, Na,Luo, Wan-Yue,Wang, Li-Li,Zhang, Ye-Yu,Wang, Jin , p. 397 - 401 (2021) |
|
COMPOUND CONTAINING INDOLEACETIC ACID CORE STRUCTURE AND USE THEREOF |
- , (2017/12/31) |
P&K INTERNATIONAL PTE. LTD. is a quality supplier of Idebenone. Our main goal is customer satisfaction. Contact us to negotiate the best price for your business on Idebenone 58186-27-9.

