Current location:Home > Markets > Pharmaceuticals


|
Manufacturing Process |
A solution containing 26.3 mg of vitamin B12 in 15 ml of water was shaken with 78 mg of platinum oxide catalyst and hydrogen gas under substantially atmospheric pressure at 25°C for 20 hours. Hydrogen was absorbed. During the absorption of hydrogen the color of the solution changed from red to brown. The solution was separated from the catalyst and evaporated to dryness in vacuo. The residue was then dissolved in 1 ml of water and then diluted with about 6 ml of acetone. After standing for several hours a small amount of precipitate (about 2 to 3 mg) was formed and was then separated from the solution. This solution was diluted with an additional 2 ml of acetone and again allowed to stand for several hours. During this time about 4 to 5 mg of noncrystalline precipitate formed. This solid was separated from the solution and an additional 2 ml of acetone was added to the solution. On standing, vitamin B12a began to crystallize in the form of red needles. After standing for 24 hours, the crystalline material was separated, yield 12 mg. By further dilution of the mother liquor with acetone additional crystalline precipitate formed (from US Patent 2,738,302). |
|
Therapeutic Function |
Hematopoietic vitamin |
|
Definition |
A form of vitamin B 12. |
InChI:InChI=1S/C62H90N13O14P.Co.H2O/c1-29-20-40-41(21-30(29)2)75(28-70-40)56-52(84)53(42(27-76)87-56)89-90(85,86)88-31(3)26-69-49(83)19-15-36-50-32(4)54-58(6,7)34(12-16-43(63)77)38(71-54)22-39-35(13-17-44(64)78)59(8,23-46(66)80)55(72-39)33(5)51-37(14-18-45(65)79)61(10,25-48(68)82)62(11,74-51)57(73-50)60(36,9)24-47(67)81;;/h20-22,28,31,34-37,42,52-53,56-57,76,84H,12-19,23-27H2,1-11H3,(H15,63,64,65,66,67,68,69,71,72,73,74,77,78,79,80,81,82,83,85,86);;1H2/q;+3;/p-3
Sterically hindered secondary alkylcobal...
(Graph Presented) We report a kinetic st...
UV-visible-NIR spectra of a variety of c...
A new coenzyme B12 (AdoCbl) analogue, 3′...
Vitamin B12, (cob(I)alamin) reduces euro...
vitamin B12s

hydroxocobalamin
| Conditions | Yield |
|---|---|
|
With
IrCl6(2-) or Fe(III)aq. or Fe(III)EDTA;
In
water;
Kinetics;
|
cob(II)alamin


hydroxocobalamin
| Conditions | Yield |
|---|---|
|
With
air;
Aerial oxidation of vitamin B12r soln. with stirring in less than 2 h at room temp.;
|

vitamin B12s

sodium hydroxide
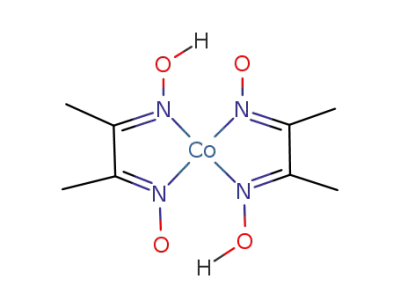
bis(dimethylglyoximato(1-))cobalt(II)
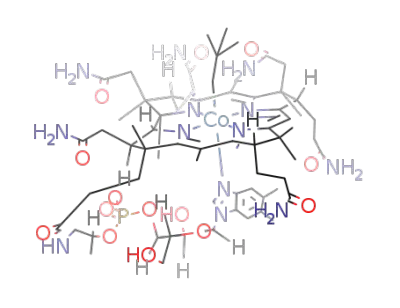
neopentylcobalamin
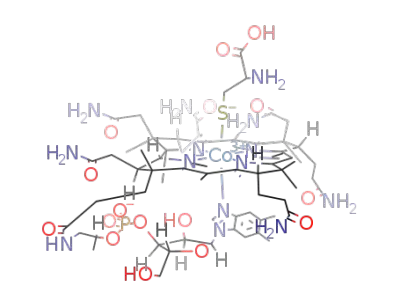
cysteinyl-vitamin B12
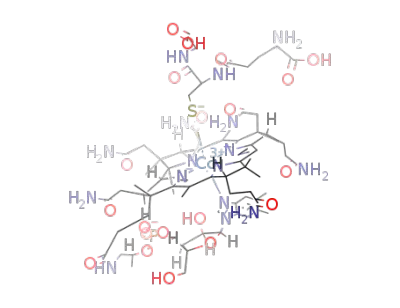
glutathionylcobalamin
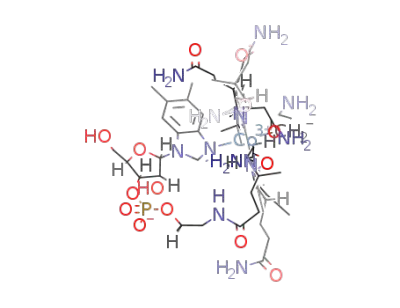
β-(3-aminopropyl)cobalamin
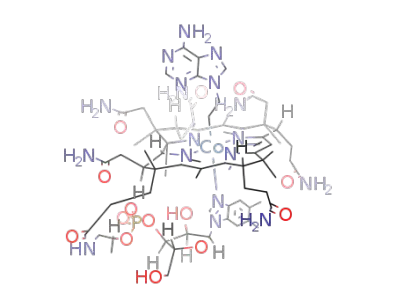
(adenylethyl)cobalamine

