Current location:Home > Markets > Nutraceuticals


|
Biological Activity |
Inhibitor of 5- and platelet 12-lipoxygenases (IC 50 values are 9.5 and 0.12 mM respectively). Also inhibits Raf-mediated MEK-1 phosphorylation in C6 rat glioma cells and induces G1 and G2 cell cycle arrest by decreasing cdk1, cdk2, cyclin D2 and cyclin A expression. Anti-inflammatory in vivo . |
|
Biochem/physiol Actions |
The flavonoid component of Nepalese and Sino-Japanese crude drugs. Baicalein, a major flavone of Scutellariae baicalensis, inhibits the 12-lipoxygenase (12-LOX) pathway of arachidonic acid metabolism, which inhibits cancer cell proliferation and induces apoptosis. |
|
General Description |
Baicalein is a flavonoid compound with potential inhibitory activity against HIV-1 integrase, targeting both the enzyme's active site and its interaction with LEDGF/p75, which may contribute to its role as an antiviral agent by disrupting viral DNA integration into the host genome. |
|
Definition |
ChEBI: A trihydroxyflavone with the hydroxy groups at positions C-5, -6 and -7. |
InChI:InChI=1/C15H10O5.H2O/c16-9-6-11(8-4-2-1-3-5-8)20-12-7-10(17)14(18)15(19)13(9)12;/h1-7,17-19H;1H2
An attempt was made to probe and elucida...
-
Five major flavonoids induced by chitin ...
The formation of the complexes of baical...
Naturally occurring polyphenolic compoun...
-
-
The invention discloses a flavonoid deri...
Biologically active plant flavonoids, in...
Angiogenesis leads to tumor neovasculari...
– A new approach to oroxylin A, a monome...
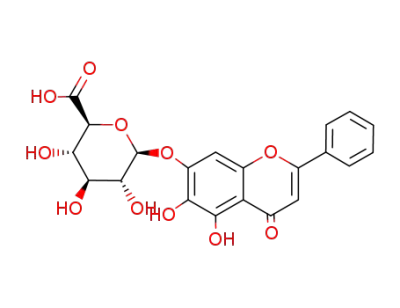
baicalin

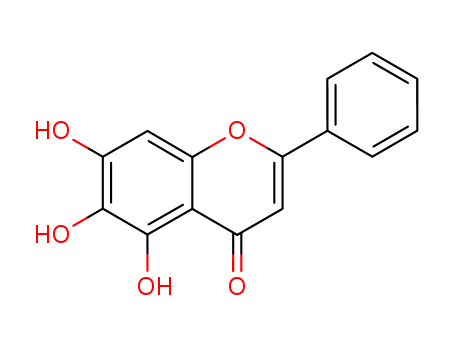
5,6,7-trihydroxy-2-phenyl-4H-1-benzopyran-4-one
| Conditions | Yield |
|---|---|
|
With
sulfuric acid; water;
at 20 ℃;
for 0.166667h;
|
93% |
|
With
sulfuric acid;
In
water;
at 90 ℃;
for 0.0833333h;
|
71.9% |
|
With
sulfuric acid;
at 90 ℃;
for 0.166667h;
|
29.3% |
|
With
hydrogenchloride;
In
ethanol; water;
for 48h;
Inert atmosphere;
Reflux;
|
19% |
|
With
sulfuric acid;
In
ethanol;
at 100 ℃;
for 8h;
Inert atmosphere;
|
10.3% |
|
hydrolysis with β-glucuronidase;
|
|
|
With
sulfuric acid; water;
for 3h;
|
|
|
baicalin;
With
sodium sulfite; Sodium carboxymethylcellulose;
at 37 ℃;
for 0.25h;
pH=7;
aq. buffer;
Enzymatic reaction;
With
β-D-glucuronidase;
at 37 ℃;
pH=7;
aq. buffer;
|
|
|
baicalin;
With
sulfuric acid; aluminium(III) chloride hexahydrate; calcium chloride;
In
ethanol; glycerol;
at 90 ℃;
for 16h;
With
phosphoric acid;
In
ethanol; glycerol;
for 0.333333h;
Sonication;
|
5.6 g |
|
With
choline chloride; urea;
In
aq. buffer;
at 40 ℃;
for 2h;
pH=5;
Temperature;
pH-value;
Solvent;
Enzymatic reaction;
|
|
|
With
hydrogenchloride; acetic acid; potassium hydroxide;
In
water;
at 90 ℃;
for 24h;
|
2.18 g |
|
With
sulfuric acid; water;
at 121 ℃;
for 0.666667h;
Temperature;
Time;
|
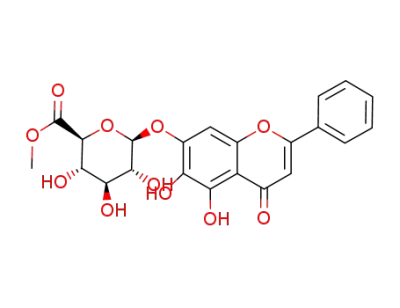
Baicalin methyl ester


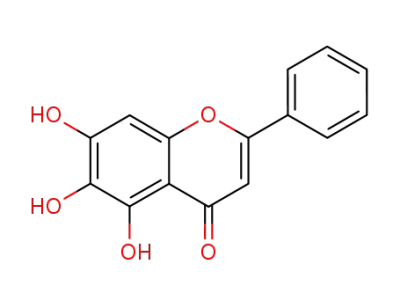
5,6,7-trihydroxy-2-phenyl-4H-1-benzopyran-4-one
| Conditions | Yield |
|---|---|
|
With
sulfuric acid;
In
ethanol; water;
at 95 ℃;
for 8.5h;
Inert atmosphere;
|
97% |
|
With
hydrogenchloride;
In
ethanol;
at 85 ℃;
for 12h;
Temperature;
Reagent/catalyst;
Inert atmosphere;
|
91.8% |
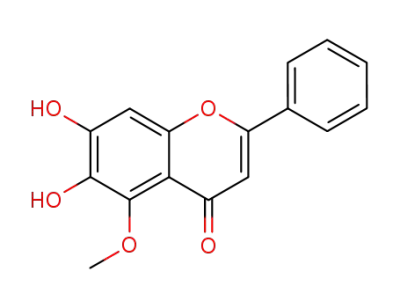
6,7-dihydroxy-5-methoxyflavone
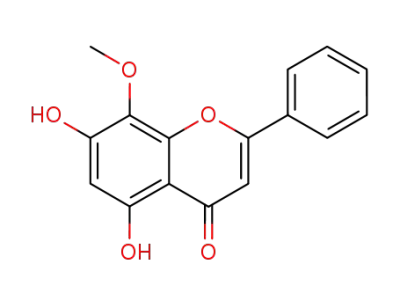
wogonin
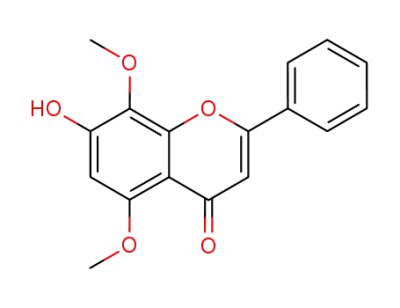
7-hydroxy-5,8-dimethoxyflavone
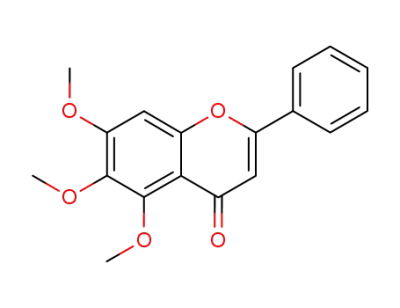
5,6,7-trimethoxyflavone

5,6,7-trimethoxyflavone
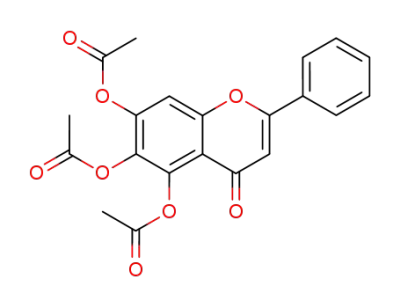
4-oxo-2-phenyl-4H-1-benzopyran-5,6,7-triyl triacetate
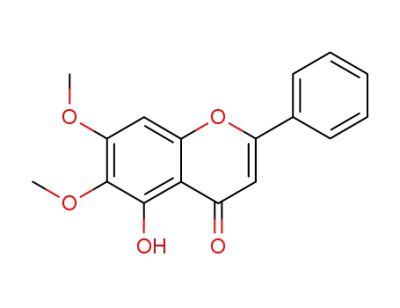
mosloflavone
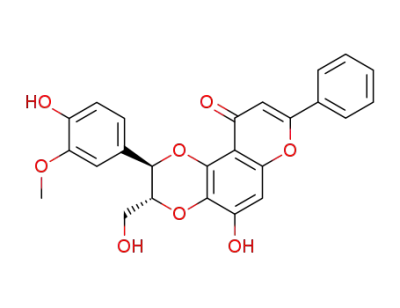
(2R,3R)-10-Hydroxy-3-(4-hydroxy-3-methoxy-phenyl)-2-hydroxymethyl-7-phenyl-2,3-dihydro-1,4,8-trioxa-phenanthren-5-one

