

|
Safety Profile |
Poison by subcutaneous andintravenous routes. Moderately toxic by ingestion. Whenheated to decomposition it emits acrid smoke and fumes.A carthartic and purgative. |
|
Purification Methods |
Aloin forms the monohydrate as lemon yellow crystals on crystallisation from H2O (450g/1.5L) which has a lower m ~70-80o than the anhydrous substance. [Cahn & Simonsen J Chem Soc 2573 1932, Hay & Haynes J Chem Soc 3141 1956.] |
|
Definition |
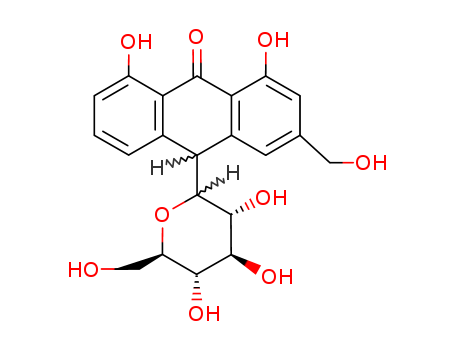
ChEBI: A C-glycosyl compound that is beta-D-glucopyranose in which the anomeric hydroxy group is replaced by a 4,5-dihydroxy-2-(hydroxymethyl)-10-oxo-9,10-dihydroanthracen-9-yl moiety (the 9S d astereoisomer). |
InChI:InChI=1/C21H22O9/c22-6-8-4-10-14(21-20(29)19(28)17(26)13(7-23)30-21)9-2-1-3-11(24)15(9)18(27)16(10)12(25)5-8/h1-5,13-14,17,19-26,28-29H,6-7H2/t13-,14?,17-,19+,20-,21+/m1/s1

10-glucopyranosyl-1,8-dihydroxy-3-(hydroxymethyl)-9(10H)-anthracenone
| Conditions | Yield |
|---|---|
|
With
ethanol; sodium ethanolate;
unter vermindertem Druck;
|
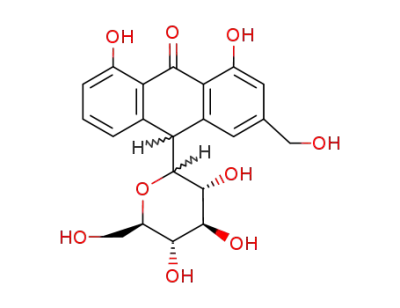
1,8-dihydroxy-3-hydroxymethyl-10-(tetra-O-acetyl-ξ-D-glucopyranosyl)-anthrone


10-glucopyranosyl-1,8-dihydroxy-3-(hydroxymethyl)-9(10H)-anthracenone
| Conditions | Yield |
|---|---|
|
With
ethanol; sodium ethanolate;
unter vermindertem Druck;
|
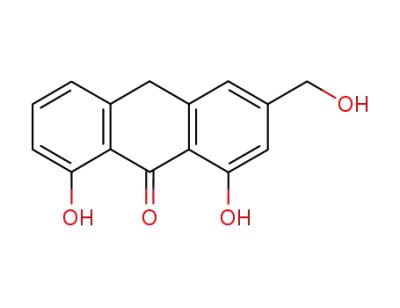
1,8-dihydroxy-3-(hydroxymethyl)-9(10H)-anthracenone
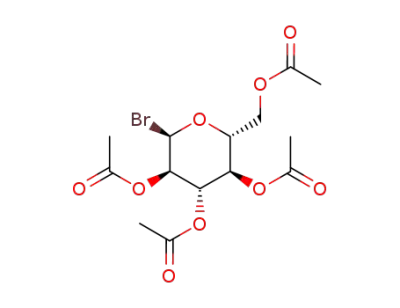
2,3,4,6-tetra-O-acetyl-α-D-glucopyranosyl bromide

1,8-dihydroxy-3-hydroxymethyl-9,10-anthracenedione
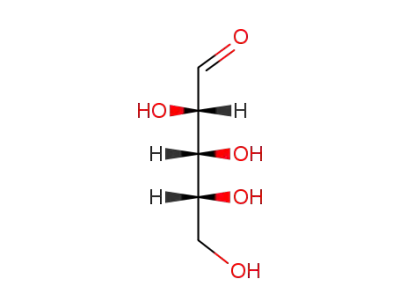
D-Arabinose

1,8-dihydroxy-3-(hydroxymethyl)-9(10H)-anthracenone
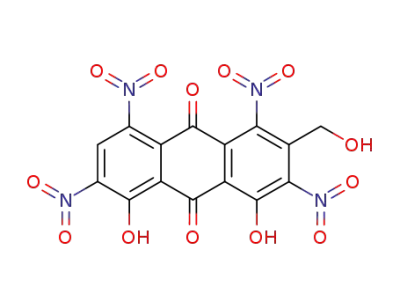
4,5-dihydroxy-2-hydroxymethyl-1,3,6,8-tetranitro-anthraquinone

