Current location:Home > Markets > Pharmaceuticals


|
Physical and Chemical Properties |
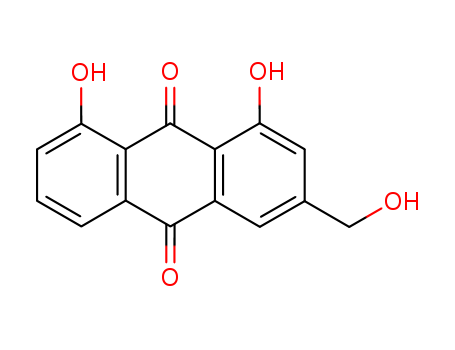
Aloe emodin aloe is one the major components of the aloe essential oils. It is either presented in aloe in the free form or presented in rhubarb, senna, and aloe in the form of glycosides. The compound precipitated from toluene or ethanol is yellow-orange needles with a relative molecular mass of 270.25 and the melting point of 223~224 ℃. It can be subject to sublimation in the carbon dioxide gas stream. It is easily soluble in hot ethanol, ether, benzene, with the solution exhibiting yellow color. It is also soluble in aqueous ammonia and sulfuric acid with the solution exhibiting crimson color. The main active ingredient of aloe is the barbaloin and aloe emodin. The aloe juice has anti-inflammatory effect and can promote healing of skin damage. The above information is edited by the lookchem of Dai Xiongfeng. |
|
Rhubarb |
Rhubarb is a kind of commonly used traditional Chinese medicine. It was initially recorded in the "Shen Nong's Herbal Classic", low-grade product. It is recorded in every generations of Ancient herbal medicine. Rhubarb is the Polygonaceae plant rhubarb Rheum, Rheum tanguticum and the dried roots or rhizomes of medicinal rhubarb Rheum officinale Baill. It is mainly produced in Sichuan, Gansu, Qinghai and other places. Rhubarb has effects such as purge heating and intestine cleaning, blood cooling and detoxification effect, and blood stasis dredging and can be applied to the treatment of the internal heating and constipation, Indigestion, abdominal pain, diarrhea, heat jaundice, vomiting blood, red eyes, swollen throat, abdominal pain, appendicitis, carbuncle swollen boils, blood stasis amenorrhea, and bruises. It can also be applied to external treatment of fire burns, upper gastrointestinal bleeding and so on. We can apply it with raw use or wine-processing, wine steaming or fried carbon. Rhubarb has inhibitory effect on most Gram-positive bacteria as well as some Gram-negative bacteria in vitro. Its main active ingredient is anthraquinone derivative with the aloe emodin, emodin and rhein yielding the best efficacy. It has inhibition on the murine leukemia P388: people extracted rhein, emodin and aloe emodin from rhubarb. These three kinds of anthraquinone can all reduce the number of cancer cells and the amount of ascites in tumor mice with different extent, in which the effect of rhein is the most significant and the effect of aloe emodin is relatively poor; the effect is generally correlated with the prolonged survival period. Emodin, rhein has a strong inhibitory effect on the biosynthesis of DNA, RNA and protein while aloe emodin strong has a weak inhibitory effect. Figure 1 Rheum officinale Baill. |
|
Definition |
ChEBI: A dihydroxyanthraquinone that is chrysazin carrying a hydroxymethyl group at position 3. It has been isolated from plant species of the genus Aloe. |
|
General Description |
Diacerein is known to be a suitable drug to treat osteoarthritis and prevent vascular diseases. It is a semi-synthetic anthraquinone derivative which acts by inhibiting the production of interleukin-1 and secretion of metalloproteases. |
|
Biochem/physiol Actions |
Laxative/cathartic compound; increases the contraction of intestinal smooth muscle by releasing endogenous acetylcholine. Anti-tumor activity is associated with an increased production of reactive oxygen species (ROS) that, in turn, reduces the mitochondrial transmembrane electrical potential, thus inducing a permeability transition that sets in motion a series of events culminating in cellular apoptosis. Genotoxicity and mutagenicity appear to be due to the inhibition of topoisomerase II activity by aloe emodin. |
InChI:InChI=1/C15H10O5/c16-6-7-4-9-13(11(18)5-7)15(20)12-8(14(9)19)2-1-3-10(12)17/h1-5,16-18H,6H2
-
-
A rapid synthetic approach to the natura...
A general method for the synthesis of na...
-
Herein, a two-step chemoenzymatic proces...
We previously reported a series of p53-e...
Herein, we report the chemoenzymatic syn...
A catalyst-free method for the synthesis...
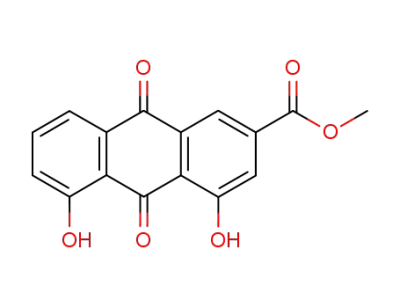
Rhein methyl ester

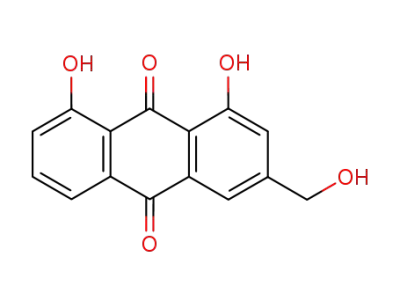
1,8-dihydroxy-3-hydroxymethyl-9,10-anthracenedione
| Conditions | Yield |
|---|---|
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 ℃;
for 2h;
Reflux;
|
65% |
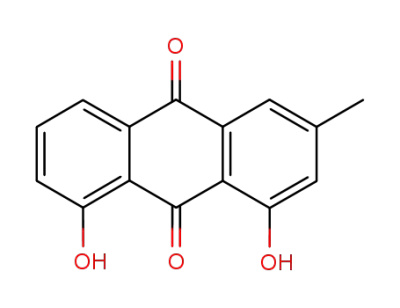
Chrysophanol


1,8-dihydroxy-3-hydroxymethyl-9,10-anthracenedione
| Conditions | Yield |
|---|---|
|
Chrysophanol;
With
N-Bromosuccinimide; dibenzoyl peroxide;
In
tetrachloromethane;
for 6h;
Heating;
With
water; calcium carbonate;
In
1,4-dioxane;
at 120 ℃;
for 7h;
|
71% |
|
Multi-step reaction with 2 steps
1: NBS; (PhCO2)2 / CCl4 / 6 h / Heating
2: 756 mg / H2O; CaCO3 / dioxane / 7 h / 120 °C
With
N-Bromosuccinimide; water; calcium carbonate; dibenzoyl peroxide;
In
1,4-dioxane; tetrachloromethane;
|
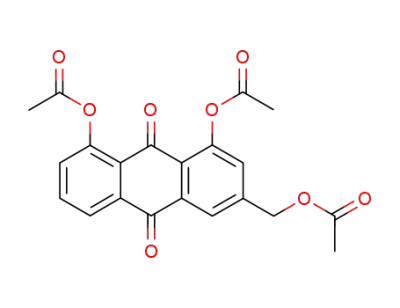
triacetyl aloe-emodin
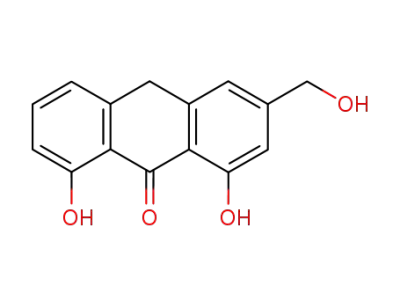
1,8-dihydroxy-3-(hydroxymethyl)-9(10H)-anthracenone
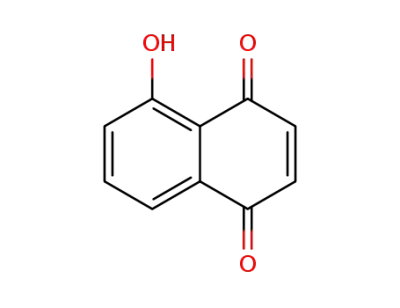
5-hydroxynaphtho-1,4-quinone
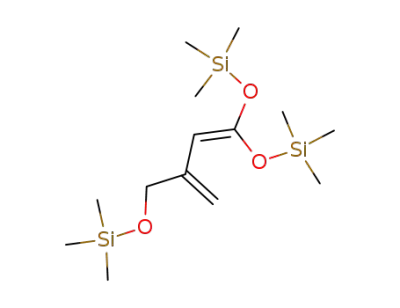
1,1-bis(trimethylsilyloxy)-3-trimethylsilyloxymethyl-1,3-butadiene

triacetyl aloe-emodin
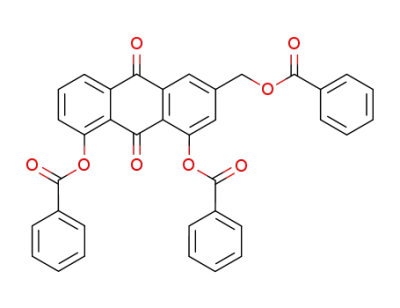
1,8-bis-benzoyloxy-3-benzoyloxymethyl-anthraquinone
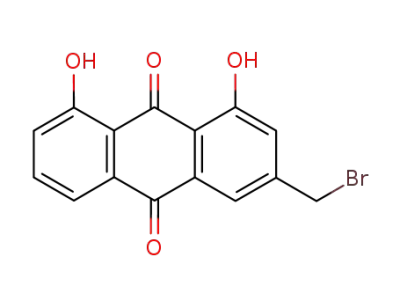
2-bromomethyl-9,10-dihydro-4,5-dihydroxy-9,10-dioxoanthracene
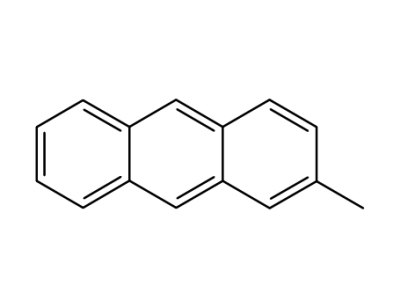
2-methylanthracene

